B-Line it to the Ultrasound Machine
Michael Schick DO, MA
CASE
A sixty-year-old male rolls in by EMS with a complaint of shortness of breath. He was discharged from prison two months ago, at which time he ceased all his medications. He believes he was on a “water-pill” for his “kidney problem” and has a “prostate problem”. He smokes a pack a day. Additionally, he complains of mild chest pain and lower extremity edema worse on the left side. He is in moderate distress, tachypneic, tachycardic, and hypoxic with three-word dyspnea. You do not appreciate any JVD, but notice decreased lung sounds at the bases with a diffuse mild expiratory wheeze. His heart is tachycardic but regular without murmurs, rubs, or gallops. His abdomen is slightly distended, but soft and non-tender. His lower extremities have 3+ edema on the left and 2+ on the right. Initial EKG is non-diagnostic.
DISCUSSION
Shortness of breath is a common chief complaint. The differential diagnosis is broad and the possibilities range from immediate life threats to those that will be managed as an outpatient. What we need is a diagnostic tool that will allow us to rapidly narrow the differential diagnosis and direct early management. Ideally this tool will be readily available and have high sensitivities and specificities for the diseases we are most concerned about. So while the x-ray tech is in another part of the emergency department and the nurse is attempting intravenous access for the third time, grab the ultrasound machine. For the patient with undifferentiated shortness of breath, ultrasound may be the best tool available to rapidly narrow the differential diagnosis and direct management.
There is useful information to be gained from the chest radiograph and it has the convenience of not requiring additional acquisition work on the part of the physician. However, ultrasound outperforms chest radiograph in nearly every situation. Ultrasound is the ideal tool to use to evaluate for pleural effusions, pericardial effusions with tamponade, and deep vein thromboses.[1] Echocardiogram can evaluate for heart failure, wall motion abnormalities, and right heart strain. For pneumothorax, chest x-ray has a sensitivity of 40% and specificity of 99% as compared to bedside ultrasound with a sensitivity of 79% and specificity of 98%.[2],[3] For pneumonia, chest x-ray has a sensitivity of 67% and specificity of 85% as compared to ultrasound with a sensitivity of 98% and specificity of 95%.[4] Additionally, ultrasound can evaluate for response of therapy in real time. Ultrasound can demonstrate improvement of pulmonary edema, resolution of a pneumothorax, as well as minute-to-minute changes in cardiac function.[5]
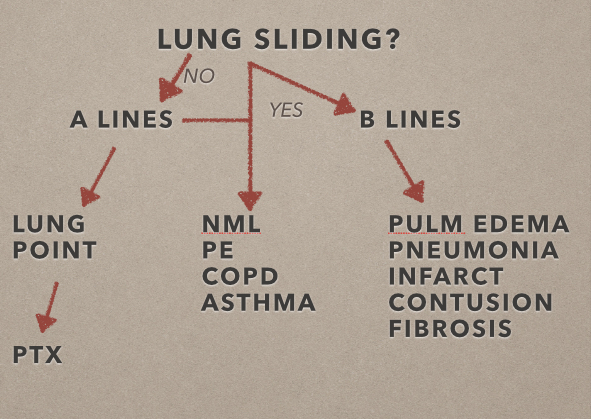
There are a number of manuscripts in the literature that describe how to use point of care ultrasound for the patient in undifferentiated shortness of breath. Lichtenstein et al. described an algorithm titled “The Blue Protocol” that can be used with lung ultrasound to differentiate shortness of breath.[6] However, the blue protocol did not include echocardiogram as part of the evaluation and for the novice sonographer the algorithm can be cumbersome. Drs. Nagdev and Mantuani demonstrated how analyzing the heart, lungs, and IVC with ultrasound can differentiate heart failure from chronic obstructive pulmonary disease (COPD).[7] Combing and simplifying these strategies creates a logical and high yield approach.
First, look at the heart. Evaluate for a pericardial effusion to rule out tamponade. If there is a pericardial effusion, view the heart in multiple windows including sub-xiphoid, parasternal long, parasternal short, and the apical four chamber to evaluate for right ventricular collapse during diastole as sonographic evidence of tamponade.[8] If you are having difficulty appreciating systole versus diastole, try using M-Mode. Point the beam over the anterior leaflet of the mitral valve in the parasternal long axis. With this image you can easily appreciate mitral valve opening during diastole as it swings up towards the inter-ventricular septum. The right ventricular free wall will be visible as a hyperechoic line between the outer anechoic pericardial effusion and inner anechoic blood within the right ventricle. When the mitral valve opens, diastole is occurring and you can easily appreciate right ventricular wall collapse into the right ventricle.
While viewing the cardiac windows gain a sense of the overall cardiac function and ejection fraction (EF). Emergency sonographers can categorize cardiac contractility as normal, depressed, or severely depressed and identify a severely depressed left ventricular EF with nearly 100% sensitivity.[9] The anterior leaflet of the mitral valve will touch or nearly touch the inter-ventricular septum in a heart with a normal EF. You may choose to measure the distance from the anterior leaflet of the mitral valve during diastole from the inter-ventricular septum with M-Mode. A distance of less than 7 mm suggests a normal EF whereas greater than 7 mm is 100% sensitive for a severely depressed EF. This is known as E-Point Septal Separation or EPSS. [10] In the apical four-chamber view, compare the right ventricle (RV) to left ventricle (LV) diameter. A normal RV is less than 2/3 the size of the LV. A dilated RV in the right setting is 98% specific of right heart strain from a pulmonary embolism. [11] You also may note a wall motion abnormality as evidence of old or new myocardial infarction. If color is added to the exam valve incompetence can be identified.
While viewing the heart in the sub-xiphoid window, point the probe’s indicator toward the patient’s head and reduce the angle of the transducer to evaluate the inferior vena cava (IVC). A pericardial effusion with a non-distensible IVC is very worrisome for tamponade. If there is respiratory variability, tamponade is excluded with few exceptions. Without a pericardial effusion, a non-distensible IVC is consistent with fluid overload states. An IVC that varies in diameter widely during respirations suggests an etiology other than heart failure.[12]
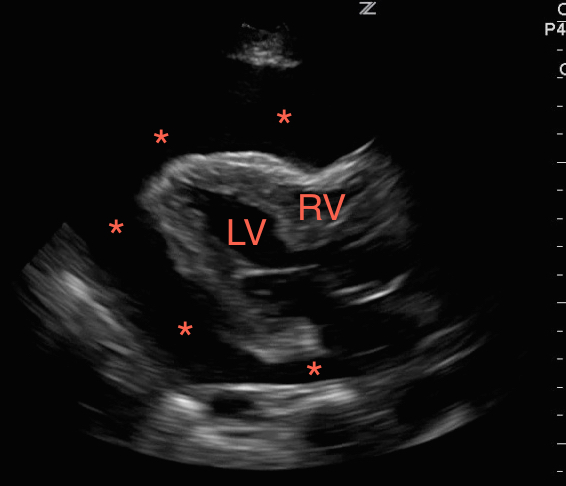
Figure 1. A large pericardial effusion (*) with tamponade physiology, right ventricular collapse during diastole.
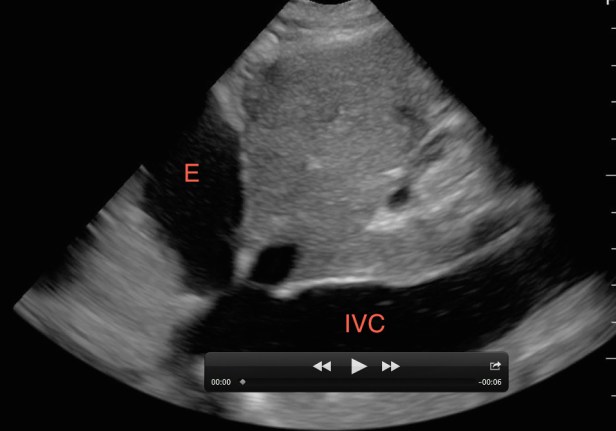
Figure 2. A non-distensible (plethoric) IVC with a pericardial effusion (E) consistent with tamponade physiology.

Figure 3. A dilated right ventricle (RV) and right atrium (RA) as compared to the left ventricle (LV) and left atrium (LA) in the setting of a pulmonary embolism.
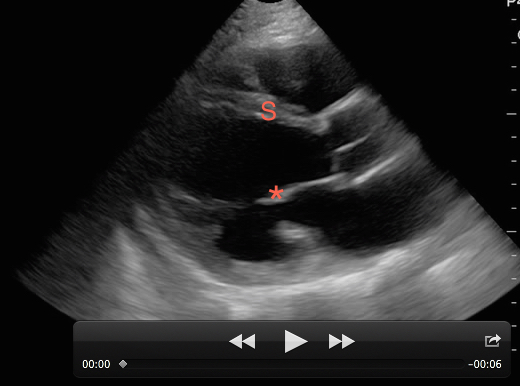
Figure 4. A poorly contracting heart in the setting of decompensated heart failure. The anterior leaflet of the mitral valve (*) does not come close to the inter-ventricular septum (S) during diastole demonstrating a severely depressed EF.
Next, move to the lungs. Fluid in the lungs will fall to the dependent portions, unlike air, which will rise to the superior and anterior chest. Start by placing the probe at each lung apex in the mid-clavicular line, 2nd-3rd intercostal space to evaluate for lung sliding. Then move to the 6th -8th intercostal spaces in the mid-axillary line to evaluate for lung sliding. In a prospective study of 27 patients, lung ultrasound was more sensitive than chest x-ray and as sensitive as computed tomography in identifying pneumothoraces. This may be done in an upright or supine position. In the setting of trauma and no lung sliding, most clinicians will assume a pneumothorax is present and place a chest tube. If the patient appears stable, look at other lung segments to find the lung point, which is the point of separation of the visceral and parietal pleura. This is nearly 100% specific for a pneumothorax. M-mode may also be used to evaluate for pneumothorax. The classic “seashore sign” is present when there is normal lung sliding, as compared to the “bar code” sign in pneumothorax. Use this for the intubated patient as well to evaluate for main stem bronchus intubation.[13]

Figure 5. With the linear array probe the pleura (P) can be seen sliding between two ribs (R) with posterior shadowing (S).

Figure 6. The seashore sign with M-mode demonstrating normal lung movement.

Figure 7. The barcode sign demonstrating a pneumothorax with M-mode.
After ruling out a pneumothorax, divide each hemi-thorax into four segments bordered by the posterior axillary line, anterior axillary line, medial clavicular line and the inter-nipple line. Evaluate the lung in each quadrant for lung sliding, A-lines, B-lines, consolidations, and effusions. A-lines are regular horizontal hyperechoic lines that reverberate from the pleural and are indicative of a normal or hyperinflated lung. If all lung segments appear to have lung sliding and A-lines then the patient’s differential is narrowed to that of a patient with a normal or hyperinflated chest x-ray. This may include asthma, COPD, anemia, acidosis, neuromuscular disorders, and pulmonary embolism. If pulmonary embolism is suspected, performing lower extremity deep vein thrombosis (DVT) studies can confirm (but not rule out) the diagnosis. In a prospective, 47 patient study a 2-point compression exam at the femoral and popliteal veins was nearly 100% sensitive and specific for DVT. [14]
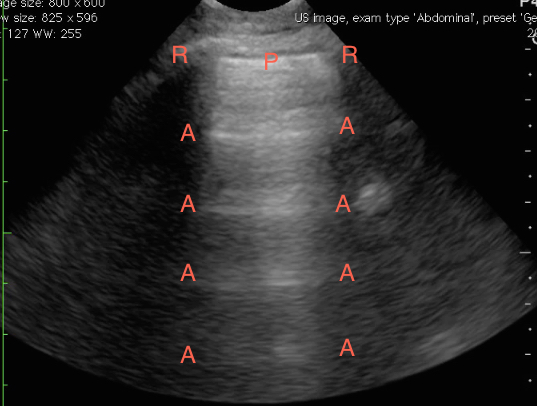
Figure 8. Normal lung ultrasound demonstrating A-lines (A) reverberating from the pleura (P) between two ribs (R).
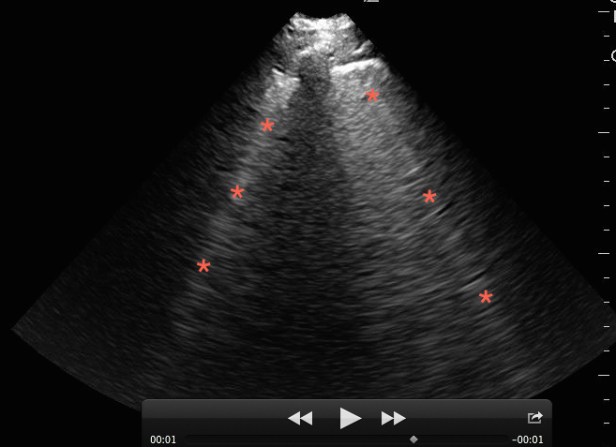
Figure 9. Lung ultrasound on a patient with pulmonary edema and B-lines (*).
B-lines are hyperechoic vertical lines that originate from the pleural line, obliterate A-lines, and often run off the bottom of the image. Three or more in the screen are indicative of interstitial syndrome. A diffuse B-line pattern in all lung segments narrows the differential to those conditions that present with a diffuse interstitial lung pattern such as pulmonary edema (cardiogenic and non-cardiogenic), multifocal pneumonia, and pulmonary fibrosis. A focal B-line pattern in one or two lung segments narrows the differential to a focal interstitial process such as pneumonia, pulmonary contusion, or pulmonary infarction.
Pleural effusions are most commonly dependent unless loculated and can most easily be visualized at the lower lung segments/costophrenic angles. Typically, above the diaphragm a normal mirror artifact of the liver or spleen can be seen, however when a pleural effusion is present there will be an anechoic pocket of fluid often with floating lung within. Additionally, the spine is not readily apparent behind the lung as air scatters the ultrasound waves. However, in the setting of a pleural effusion the ultrasound waves are transmitted well and the spine may be apparent. This is called the “spine sign”. Consolidated lung appears like solid tissue on ultrasound and is often referred to as “hepatization” of lung as it has a similar appearance to the liver.[15]

Figure 10. A small pleural effusion (*) visualized above the diaphragm (D) with floating lung (L) and posterior spine sign (S).

Figure 11. A pulmonary consolidation (C) with a diffuse B-line pattern (B) visualized above the liver (L) and diaphragm (*).
Combining the cardiac, IVC, lung, and DVT (when indicated) point of care ultrasound exams rapidly narrows the differential diagnosis and results in early directed management. The use of point of care ultrasound results in more time at the bedside, but the initial investment of time is well worth it when you catch important pathology, make critical diagnoses early, and save lives. Multiple studies demonstrate the relative ease and short period of time it takes to use these techniques effectively.[16] However, even expert sonographers should understand their limitations and not hesitate to employ other diagnostic tools when necessary. The largest limitations of point of care ultrasound are the operator’s expertise, experience, and the patient’s body habitus.
CASE RESOLUTION
The patient had a markedly decreased EF, dilated IVC, diffuse B-line pattern on lung exam, and negative DVT studies. Additionally, he had bilateral moderate pleural effusions and a modest amount of abdominal free fluid. His clinical picture was consistent with congestive heart failure and improved with BiPAP, nitroglycerin, and diuretics. He was admitted to the cardiology service and was discharged two days later on a new medication regiment.
References
[1] Mavrizio et al. Can chest ultrasonography replace standard chest radiography for evaluation of acute dyspnea in the ED? CHEST (2011) 39(5):1140-7
[2] Alrjab et al. Pleural ultrasonography versus chest radiography for diagnoses of pneumothorax: review of the literature and meta-analysis. Crit Care (2013) 17:R208
[3] Ding et al. Diagnosis of Pneumothorax by radiography and ultrasonography a meta-analysis. CHEST (2011) 140(4):859-66
[4] Cortellaro et al. Lung ultrasound is an accurate diagnostic tool for the diagnosis of pneumonia in the emergency department. Emerg Med J (2012) 29(1):19-23
[5] Noble V. Ultrasound assessment for extravascular lung water in patients undergoing hemodialysis. Time course for resolution. CHEST (2009) 135:1433-39
[6] Lichtenstein DA, Mezie`re GA: Relevance of lung US in the dx of acute respiratory failure: The BLUE protocol. CHEST (2008) 134(1):117–125
[7] Mantvani and Nagdev. Three-view bedside ultrasound to differentiate cute decompensated heart failure from COPD. Am J Emerg Med 2013
[8] Nagdev and Stone. Point-of-care ultrasound evaluation of pericardial effusions: Does the patient have cardiac tamponade? Resuscitation (2011) 82:671-673
[9] Anderson et al. Diagnosing heart failure among acutely dyspneic patients with cardiac, inferior vena cava, and lung ultrasonography. Am J Emerg Med 2013
[10] McKaigney et al. E-point septal separation: a bedside tool for emergency physician assessment of the left ventricular ejection fraction. Am J of Emerg Med 2014
[11] Dresden et al. Right Ventricular Dilatation on Bedside Echocardiography Performed by Emergency Physicians Aids in the Diagnosis of Pulmonary Embolism. Annals of Emerg Med (2014) 63:16-24
[12] Anderson et al 2013
[13] Rowan et al. Traumatic Pneumothorax Detection with Thoracic US: Correlation with Chest Radiography and CT-Initial Experience. Radiology (2002): 210-214
[14] Crisp et al. Compression Ultrasonography of the Lower Extremity With Portable Vascular Ultrasonography Can Accurately Detect Deep Venous Thrombosis in the Emergency Department. Annals of Emerg Med (2010) 56(6):601-610
[15] Volpicelli et al. International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med (2012) 38:577-591
[16] Noble et al. Evaluation of a thoracic ultrasound training module for the detection of pneumothorax and pulmonary edema by prehospital physician care providers. BMC Med Educ. (2009) 12;9:3

